Resource efficient catalysis
Novel organocatalysts and cooperative catalytic procedures for the utilization of CO2 as a synthetic building block
Various studies suggest a close connection between global climate change and the emission of anthropogenic greenhouse gases. By far largest part of this emission is attributed to carbon dioxide (CO2). Apart from the goal of reducing CO2 emissions, its use as synthetic building block is the central point of the overall CO2 management strategy. The atom economic and efficient utilization of CO2 as synthetic building block is closely connected to the effective activation of this very stable molecule.
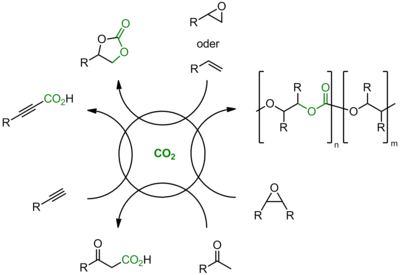
The aim of our work is the development of novel metal free catalysts, so called organocatalysts, for the synthesis of industrially relevant products with CO2 as a C1-building block. The aim is that combining those catalysts with metal-catalyzed or enzyme-catalyzed procedures in (sequential) one pot reactions leads to innovative and sustainable catalytic systems with high selectivity and energy efficiency respectively. These alternative methods, taking steps in the upstream and downstream phases, are targeted at changing and extending the raw material base, utilizing CO2. Subjects under study include transformations and products of much interest to industry and large CO2-fixation potential (Figure 1).
Below a short outline about two selected sub-projects will be given:
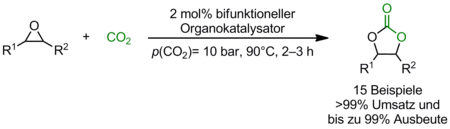
So far we prepared a series of bifunctional phosphonium salts and evaluated their potential as catalysts under various reaction conditions in the synthesis of cyclic carbonates from epoxides and carbon dioxide. Based on this catalyst screening we determined structure activity relationships and identified promising candidates. Moreover, the reaction parameters were optimized for a model reaction. Even under mild conditions (90°C, p(CO2)= 10 bar) quantitative conversions were obtained after 2–3 h for 15 different epoxides (Scheme 1). The reaction was also performed on a multi gram scale and monitored by in-situ FTIR.
Synthesis of polycarbonates
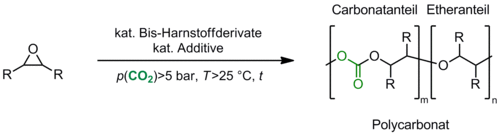
Bis-urea derivatives are known as oxo-anion receptors. We prepared a series of those compounds and employed them as catalysts in the copolymerisation of CO2 with epoxides (Scheme 2). The intention in using the urea derivatives was to stabilize the anionic intermediates in polymer chain growth and thus to support the incorporation of carbon dioxide into the polymer chain. The aim is for this to lead to a polymer with well-defined structure and material characteristics. We obtained copolymers with >80% carbonate linkage and molecular weights of >35,000 g·mol–1 in excellent yields: results depended on the catalytic system and the reaction conditions.

This project is funded by the Federal Ministry of Education and Research (BMBF) within the funding initiative "Technologies for Sustainability and Climate Protection – Chemical Processes and Use of CO2".
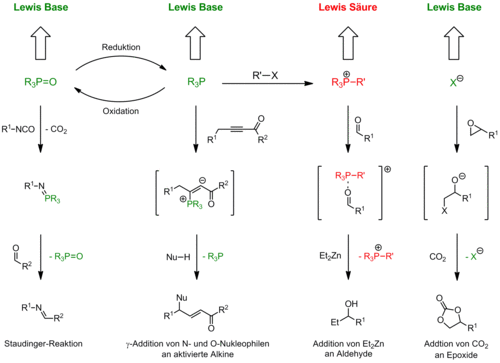
Phosphines are an important class of ligands in metal complexes. Due to their outstanding role in homogeneous catalysis, there have been numerous publications reported concerning the synthesis of achiral as well as chiral phosphine-based ligands. In addition many of them are commercially available. However phosphines and their derivatives are not only employed as ligands but are also important reagents in organic synthesis. For example, they mediate the conversion of alcohols to halides (Appel reaction), the olefination of ketones and aldehydes (Wittig reaction) and the synthesis of imines via Staudinger reaction. In contrast to their tremendous importance as ligands and reagents, the application of phosphines as organocatalysts typically has a somewhat secondary role.
We are exploring the application of phosphorus-based organic compounds as catalysts in a variety of reactions. Therefore we are utilizing their Lewis basic and Lewis acidic properties, respectively (Figure 2). On the one hand our aim is to develop asymmetric versions of the reactions indicated, by employing chiral phosphines or derivatives. A second aim is to develop catalytic (asymmetric) variations of methods which hitherto employ phosphines in stoichiometric amounts, e.g. Wittig Reactions.
A short outline of selected projects is given below:
Asymmetric intramolecular γ‑addition to activated alkynes
A variety of O-, S-, and N-functionalized activated alkynes were prepared and cyclised in the phosphine catalyzed intramolecular γ-Addition (Scheme 3). For the first time we successfully converted N‑derivatives in this reaction. In an intense screening of chiral phosphine catalysts products with up to 84% ee were obtained in an intense screening of chiral phosphine catalysts.

Addition von Et2Zn to aldehydes
Alongside the application of phosphines as Lewis basic catalysts we are also interested in the utilization of phosphonium salts as potential Lewis acidic organocatalysts. In this context we studied the addition of diethyl zinc to aldehydes (Scheme 4). It is known that this reaction can be catalyzed by Lewis acids. We showed that simple Bu4PCl is an efficient catalyst for this reaction. However, when chiral phosphonium salts were used, only racemic products were obtained and it was observed that the anion exerted a strong influence. Further investigations revealed that diethyl zinc is activated by the Lewis basic counter ion and the reaction can be catalyzed by a combination of simple alkaline metal salts and crown ethers.
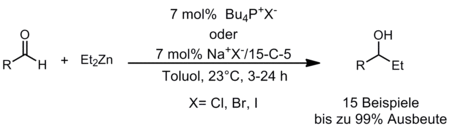
Catalytic Wittig reaction
In our efforts to convert phosphine mediated processes into catalytic variants we investigated the Wittig reaction. So far we have developed a microwave-assisted catalytic variant employing commercially-available tributyl phosphineoxide, as well as a thermal method utilizing an easily-accessible phospholane oxid as a pre-catalyst (Scheme 5). The right combination of reducing agent, base and solvent recently enabled us to convert over 20 substrates. In some cases yields and selectivity levels were significantly higher than previously reported.

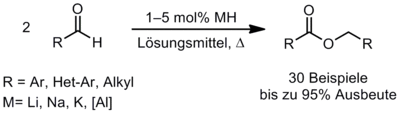
Metall-Hydrid-Mediated Tishchenko-Reaction
During our studies concerning the catalytic Wittig reaction we observed the dimerization of benzaldehyde yielding benzyl benzoate (Tishchenko reaction) when sodium hydride was used as the base in our model reaction. Based on this result we developed an efficient method for the dimerization of aromatic aldehydes (Scheme 6). The reaction was also produced on a large scale and easily monitored by in situ FT-IR spectroscopy. A mechanism was postulated and confirmed by labelling and capture experiments. Thus, we developed an efficient method for the dimerization of aromatic as well as hetero-aromatic and aliphatic aldehydes.