Catalyst design for electrosynthesis
Dr. Bernd Müller
One way to carry out electrochemical syntheses is to use homogeneous catalysts ("mediators"). These are redox-active species that can be regarded as electrochemically regenerable reagents. Electron transfer first takes place between the electrode and the catalyst under formation of the active species. The latter reacts with a substrate, which then converts to the desired product. This indirect electron transfer usually allows large overpotentials to be avoided and selectivities to be controlled. The mediator as electron carrier is always recovered at the electrode and is not consumed during electrosynthesis. The reaction of the mediator with the substrate takes place in homogeneous phase.
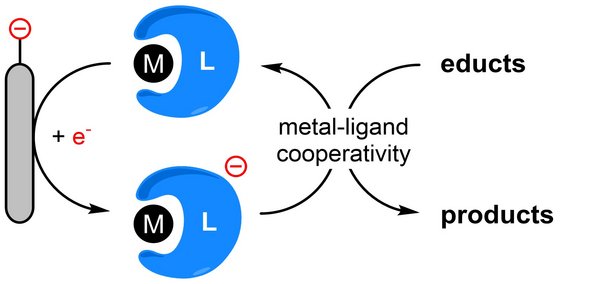
The electrode material usually plays a major role in the design of electrosyntheses. However, in mediated electrochemistry, the electrodes are no longer the site of the primary substrate reaction. Analogously to classical homogeneous catalysis, the mediator can be tailored via the choice of transition metal and ligand structure to meet the needs of the desired reaction. This expands the possibilities of electrosynthesis in terms of selectivity and possible reaction mechanisms.
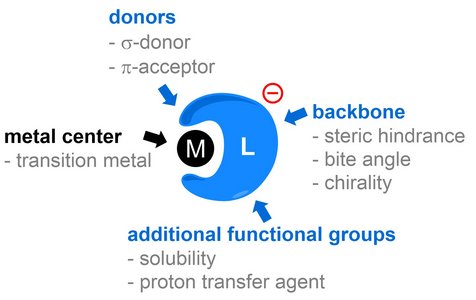
The aim is to explore the mechanisms of already known mediated electrosyntheses and to develop new syntheses that follow this design principle to the point of application maturity.
[1] R. Francke, M. Roemelt, B. Schille, Chem. Rev. 2018, 118, 4631–4701.
[2] L. F. T. Novaes, J. Liu, Y. Shen, L. Lu, J. M. Meinhardt, S. Lin, Chem. Soc. Rev. 2021, 50, 7941-8002.

