Catalysis of late transition metals
Prof. Torsten Beweries
The group is currently working on the fundamentals of catalytic reactions involving late transition metal complexes on the basis of preparative organometallic chemistry. The aim is to gain insights into the development of new and optimisation of known catalytic processes by studying structure-activity relationships. In general, the focus is on various aspects of the activation and transformation of small, partially unreactive molecules such as alkenes, alkynes, H2O, N2, CO2, or H2. Another major research field of the group is the dehydrocoupling of main group compounds such as amine-boranes for the synthesis of new potentially catalytically active inorganic materials. One of the group's strengths is its extensive experience in the field of preparative organometallic chemistry and coordination chemistry. This is complemented by the expertise available in the group and in the research area in the field of mechanistic investigation of catalytic processes.
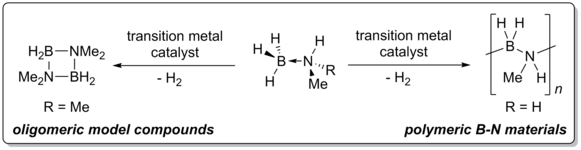
In recent years, the catalytic formation of bonds between main group elements has often been investigated, e.g., in the context of hydrogen storage in amine-borane adducts or in connection with the development of new inorganic materials.1 Compared to the formation of isovalence electronic C-C bonds, the synthetic potential of the formation of B-N bonds and B-N compounds themselves has nevertheless been comparatively little investigated. Amine-borane adducts are suitable as precursors for the synthesis of new B-N materials. In our group, we work on the transition metal complex-catalysed dehydrocoupling of amine-borane adducts such as methylamine-borane and its substituted and functionalised analogues for the synthesis of new oligomeric and polymeric B-N materials.
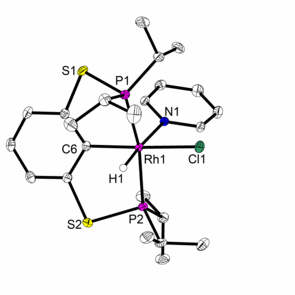
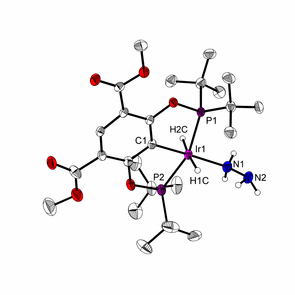
Catalysts used for these reactions include complexes with classical PCP and PNP ligands, but also novel PNP and PBP pincer complexes, which are developed in the group and systematically investigated with regard to their coordination chemistry and applications in catalysis, e.g., in selective hydrogenations.3
The group uses standard equipment for working under inert conditions (high-vacuum Schlenk lines, gloveboxes), electrochemical equipment, fully automated volumetric instruments for measuring gas evolution and gas consumption, which can also be operated under photochemical conditions, autoclave equipment, as well as gas chromatography, 31P benchtop NMR spectroscopy, mass spectrometry and UV-vis spectroscopy.
Students interested in organometallic synthesis, ligand design and the application in homogeneous catalysis are welcome to join the group as part of internships, bachelor and master theses or PhD projects.
Please send an email to torsten.beweries{at}catalysis.de.
References
[1] | a) T. Beweries, H. Helten, in Encyclopedia of Inorganic and Bioinorganic Chemistry, 2020, pp. 1-25; b) D. Han, F. Anke, M. Trose, T. Beweries, Coord. Chem. Rev. 2019, 380, 260-286. |
| [2] | a) P. Hasche, J. Haak, F. Anke, C. Kubis, W. Baumann, H. J. Drexler, H. J. Jiao, T. Beweries, Catal. Sci. Technol. 2021, 11, 3514-3526, b) F. Anke, S. Boye, A. Spannenberg, A. Lederer, D. Heller, T. Beweries, Chem. Eur. J. 2020, 26, 7889-7899; c) R. Knitsch, D. Han, F. Anke, L. Ibing, H. Jiao, M. R. Hansen, T. Beweries, Organometallics 2019, 38, 2714-2723; d) F. Anke, D. Han, M. Klahn, A. Spannenberg, T. Beweries, Dalton Trans. 2017, 46, 6843-6847; e) D. Han, M. Joksch, M. Klahn, A. Spannenberg, H. J. Drexler, W. Baumann, H. Jiao, R. Knitsch, M. R. Hansen, H. Eckert, T. Beweries, Dalton Trans. 2016, 45, 17697-17704. |
| [3] | a) D. Decker, Z. Wei, J. Rabeah, H.-J. Drexler, A. Brückner, H. Jiao, T. Beweries, Inorg. Chem. Front. 2022, 9, 761-770; b) M. Joksch, H. Agarwala, M. Ferro, D. Michalik, A. Spannenberg, T. Beweries, Chem. Eur. J. 2020, 26, 3571-3577; c) P. Hasche, A. Spannenberg, T. Beweries, Organometallics 2019, 38, 4508-4515; d) G. Vlahopoulou, S. Möller, J. Haak, P. Hasche, H. J. Drexler, D. Heller, T. Beweries, Chem. Commun. 2018, 54, 6292-6295. |