Catalysis of Early Transition Metals
Dr. Fabian Reiß
The group investigates the fundamentals of catalysts of early transition metal complexes, especially of group 4 metals, based on preparative organometallic chemistry and coordination chemistry. The focus of the research is on the coordination and activation of small unsaturated as well as inert molecules (e.g. olefins, alkynes, isocyanides, nitriles, carbodiimides, CO2), which can be transformed into sometimes unusual organometallic and organic structures by coupling to metal complexes in stoichiometric and catalytic reactions.1
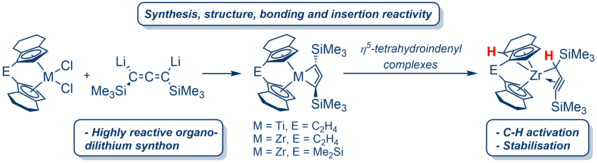
We are exploring the chemistry of unusual group 4 metallacycles with the goal of pushing the limits of coordination chemistry. The incorporation of unsaturated units into cyclic structures increases the ring strain, which limits the feasibility of such structures. Nevertheless, the formal substitution of, for example, methylene units against suitable metal fragments offers access to several exotic complexes. Thus, after testing various approaches and optimizing the synthesis of a suitable organo-dilithium synthone, we succeeded in preparing for the first time a titanacyclobuta-2,3-diene, which formally corresponds to an organometallic analogue of the highly strained and non-existent cyclobuta-1,2-diene.2 We have shown that the ansa-metallocene ligand applied, significantly contributes to the stabilization of this unusual structural motif. Recently, we were able to transfer this concept to zirconocenes where the zirconacyclobuta-2,3-dienes undergo a previously unexplored C-H activation at the ansa-ligand and form novel propargyl/allenyl complexes.3 Currently, detailed studies of the reactivity of this new class of compounds towards small unsaturated or inert molecules are underway.
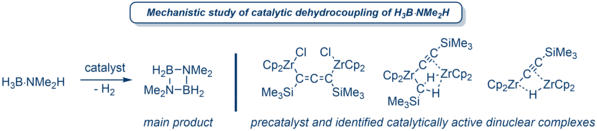
When the metallocene is changed to Cp2MCl2 (M = Zr, Hf), the organo-dilithium synthon reacts either to unusual eight-membered metallacyclooctatetraenes or to dinuclear allene bridged metallocene-chlorido complexes, depending on the stoichiometry used.2b,4 We intensively investigated the latter as catalysts for the dehydrocoupling of amine-borane adducts in a cross-disciplinary project, using experimental findings as well as quantum mechanical calculations to reveal the dinuclear nature of the catalyst as a key feature.5 Based on extensive knowledge of the chemistry of group 4 metallocene complexes, new catalyst structures are currently being developed and tested in further investigations, and the existing systems are being investigated in further catalytic dehydrocouplings.
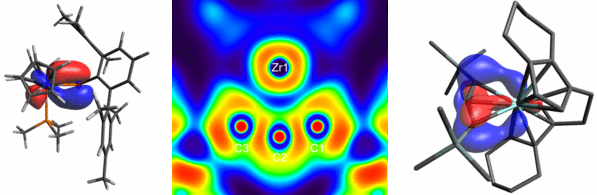
In recent years, we have also been able to establish modern methods of quantum mechanical modelling of catalyst systems in the group. The systematic analysis of proposed reaction mechanisms with the help of density functional theory calculations supports our experimental findings in an excellent way and generates positive feedback for future catalyst development.5b A series of quantum mechanical bond analyses, e.g. NBO, AIM, CAS helps us to gain a deeper understanding of the structure-property relationships in active catalyst-substrate complexes.6
The group uses standard equipment for working under inert conditions (high-vacuum Schlenk lines, gloveboxes), electrochemical equipment, fully automated volumetric instruments for measuring gas evolution and gas consumption, which can also be operated under photochemical conditions, autoclave equipment, as well as gas chromatography, 31P benchtop NMR spectroscopy, mass spectrometry and UV-vis spectroscopy.
Students interested in organometallic synthesis, ligand design and the application in homogeneous catalysis are welcome to join the group as part of internships, bachelor and master theses or PhD projects.
Please send an email to torsten.beweries{at}catalysis.de if you are interested.
[1] a) F. Reiß, K. Altenburger, D. Hollmann, A. Spannenberg, H. Jiao, P. Arndt, U. Rosenthal, T. Beweries, Chem. Eur. J. 2017, 23, 7891; b) M. Reiß, F. Reiß, A. Spannenberg, P. Arndt, T. Beweries, Organometallics, 2018, 37, 4415; c) P. Arndt, M. Reiß, A. Spannenberg, C. Schünemann, F. Reiß, T. Beweries, Dalton Trans. 2019, 48, 16525.
[2] a) F. Reiß, M. Reiß, A. Spannenberg, H. Jiao, D. Hollmann, P. Arndt, U. Rosenthal, T. Beweries, Chem. Eur. J. 2017, 23, 14158; b) F. Reiß, M. Reiß, A. Spannenberg, H. Jiao, W. Baumann, P. Arndt, U. Rosenthal, T. Beweries, Chem. Eur. J. 2018, 24, 5667; c) F. Reiß, M. Reiß, J. Bresien, A. Spannenberg, H. Jiao, W. Baumann, P. Arndt, T. Beweries, Chem. Sci. 2019, 10, 5319.
[3] X. Shi, S. Li, M. Reiß, A. Spannenberg, T. Holtrichter-Rossmann, F. Reiß, T. Beweries, Chem. Sci. 2021, 12, 16074.
[4] K. Lindenau, E. Zander, C. Schünemann, A. Spannenberg, M. V. Andreev, V. V. Burlakov, F. Reiß, T. Beweries, Organometallics 2021, 40, 3177.
[5] a) M. Trose, M. Reiß, F. Reiß, F. Anke, A. Spannenberg, S. Boye, A. Lederer, P. Arndt, T. Beweries, Dalton Trans. 2018, 47, 12858; b) K. Lindenau, N. Jannsen, M. Rippke, H. Al Hamwi, C. Selle, H. J. Drexler, A. Spannenberg, M. Sawall, K. Neymeyr, D. Heller, F. Reiß, T. Beweries, Catal. Sci. Technol. 2021, 11, 4034.
[6] a) A. Schumann, F. Reiß, H. Jiao, J. Rabeah, J. E. Siewert, I. Krummenacher, H. Braunschweig, C. Hering-Junghans, Chem. Sci. 2019, 10, 7859; b) M. Fischer, F. Reiß, C. Hering-Junghans, Chem. Comm. 2021, 57, 5626; c) F. Reiß, A. Villinger, H. Brand, W. Baumann, D. Hollmann, A. Schulz, Chem. Eur. J. 2022, DOI: 10.1002/chem.202200854.