Photochemistry and Photocatalysis
Associated Research Group Prof. Malte Brasholz (University Rostock)
Overview
Photochemistry methods allow the synthesis of some interesting product structures, which are not accessible by conventional thermal reaction control. Modern photocatalysis harnesses the classical elementary steps of photochemical transformations in catalytic processes. These include photoelectron transfer (PET) reactions, atom and group transfer reactions, and processes induced by energy transfer. Furthermore, reactive oxygen species can be photocatalytically generated for green oxidation of organic substrates. Our group focuses on the development of new synthetic methods in the field of photochemistry and photocatalysis, with different key areas.
Photoredox catalysis allows for the generation of C- and N-centered radicals under mild reaction conditions and starting from readily available radical precursors. Catalytic transition metal species such as ruthenium(II) and iridium(III) complexes, as well as a number of organic photocatalysts, exhibit favorable excited state redox potentials to activate a wide range of organic reactants in one-electron oxidations or reductions. A particular interest is in reductive radical additions and radical cascades, which allow the dearomatization of aromatic substrates.
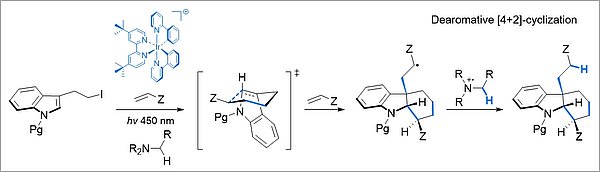
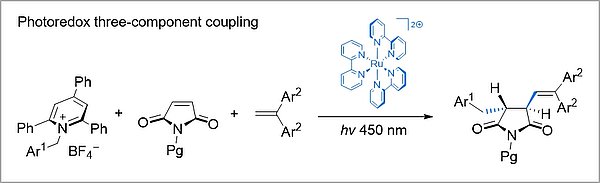
Radical multicomponent reactions can also be initiated by photoelectron transfer. In these processes, value-added polyfunctionalized reaction products are efficiently obtained from simple starting materials.
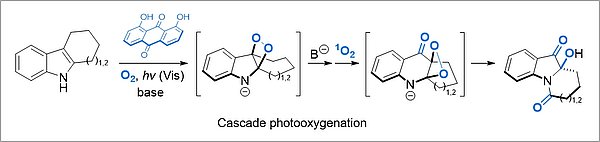
Selective catalytic photooxygenation of organic substrates leads to synthetically highly valuable primary products that are suitable as building blocks in natural product synthesis, or exhibit useful biological properties. The photooxygenation of N-heterocyclic compounds is the main focus here. Likewise, transformations of biogenic compounds and compounds derived from renewable resources are investigated.
For more information, please visit our University hompage.
Literature:
1. Tandem Organocatalysis and Photocatalysis: An Anthraquinone-Catalyzed Indole-C3-Alkylation/Photooxidation/1,2-Shift Sequence, Stephanie Lerch, Lisa-Natascha Unkel, Malte Brasholz, Angew. Chem. Int. Ed. 2014, 53, 6558-6562.
2. A Photoredox-Induced Stereoselective Dearomative Radical (4+2)-Cyclization/1,4-Addition Cascade for the Synthesis of Highly Functionalized Hexahydro-1H-carbazoles, Dirk Alpers, Malte Gallhof, Julian Witt, Frank Hoffmann, Malte Brasholz, Angew. Chem. Int. Ed. 2017, 56, 1402-1406.
3. Visible light‐mediated aerobic tandem dehydrogenative Povarov/aromatization reaction: synthesis of isocryptolepines, Eva Schendera, Lisa-Natascha Unkel, Phung Phan Huyen Quyen, Gwen Salkewitz, Frank Hoffmann, Alexander Villinger, Malte Brasholz, Chem. Eur. J. 2020, 26, 269-274.
4. Visible light cascade photooxygenation of tetrahydrocarbazoles and cyclohepta[b]indoles: access to C,N‐diacyliminium ions, Mario Frahm, Thorsten von Drathen, Lisa Marie Gronbach, Alice Voss, Felix Lorenz, Jonas Bresien, Alexander Villinger, Frank Hoffmann, Malte Brasholz, Angew. Chem. Int. Ed. 2020, 59, 12450-12454.
5. Photoredox-induced deaminative radical-cationic three-component couplings with N-alkylpyridinium salts and alkenes, Paul Seefeldt, Rajesh Dasi, Alexander Villinger, Malte Brasholz, ChemPhotoChem 2021, 5, 979-983.
