Catalysis for Sustainable Syntheses
Prof. Jagadeesh Rajenahally
The world has transformed significantly in the last and current century with notable advancements as a result of scientific discoveries and their applications. Among these, chemical research plays an important role in the progression of sustainable society with respect to health, energy and environment as well as economy. In particular, chemical synthesis (organic synthesis) occupies a unique position in chemistry, the central science, with a profound impact on the modernization of society and human lives. Since the first laboratory synthesis of an organic molecule, urea, in the year 1828 by the German Chemist Friedrich Wöhler, chemical synthesis is gaining increasing importance and emerged as an indispensable tool to produce of all kinds of compounds, pharmaceuticals, agrochemicals, fuels, materials and other daily life products. One of the major goals of chemical research is to develop more sustainable processes for advanced organic synthesis as well as effective valorization of renewable feedstocks. Notably, catalysis constitutes a key technology, which is decisive to achieve chemical processes in a more efficient, sustainable, and environmentally friendly manner. Catalysis governs the chemical synthesis and occupies a major place in both research laboratories and industries with extensive applications in the production of all kinds of chemicals and life science molecules as well as daily life products. Thus, the modern chemical synthesis and industrialized world mainly relies on catalysis and would be inconceivable without the applicability of catalytic processes. As a result, >80% of globally produced chemicals are made using catalytic processes. In general, the design of ‘‘ideal’’ and practical catalysts is of prime importance and crucial for the production of all kinds of chemicals and life science molecules at present and in the future.
Our research focuses on:
(1) Development of inexpensive, durable and practical catalysts with the activity and selectivity of the homogeneous ones, and the stability and recyclability of the heterogeneous ones.
(2) Development of more sustainable and environmentally beneficial catalytic processes for the synthesis of fine and bulk chemicals, pharmaceuticals and agrochemicals as well as for the valorization of renewable feedstocks.

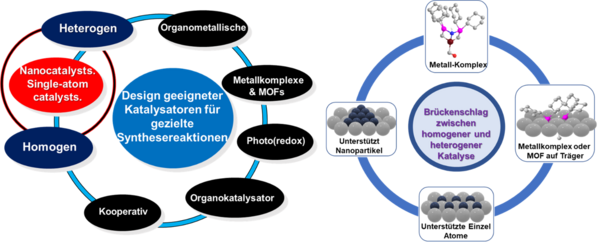
We focus on the development of both homogeneous and heterogeneous catalysts for organic synthesis. Homogeneous complexes are active and highly selective but not very stable and are difficult to recycle or reuse. On the other hand, heterogeneous materials are quite stable and can be conveniently recyclable, but these ones exhibit less selectivities and activities. Thus, the major challenge in catalysis is to create catalysts, which exhibit more stability with convenient recycling, high activities, and remarkable selectivities. To design such ‘ideal’ catalysts, combining both homogeneous and heterogeneous catalysis aspects is considered to be a promising approach. In this regard, our group is dealing on how to combine both homogeneous and heterogeneous catalysis to create ‘break through’ catalysts to bridge the gap between both of these aspects. Interestingly the immobilization and pyrolysis of metal complexes or metal organic frameworks (MOFs) on heterogeneous supports emerging as a potential methodology for the preparation of supported nanoparticles (NPs)- and single atom (SACs)-based materials, which are considered to be mimic both heterogeneous and homogeneous catalysis features. Applying this approach, we successfully prepared 3d metal-based NPs and SACs, which constitute highly active and selective as well as stable and reusable catalysts for advanced organic synthesis involving functionalized and structurally diverse molecules.
Our selected publications related to these works: Science, 2013, 342, 1073-1076; Science, 2017, 358, 326-332; Nature Catal., 2022, 5, 20; Nature Chem., 2013, 5, 537-543.
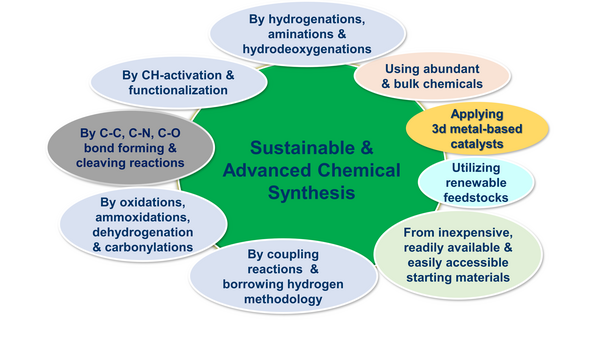
Organic synthesis is an imperative tool that has the power to replicate the fascinating molecules of living nature in the laboratory and industry. These synthetic strategies allow to produce chemical products, materials and life science molecules for potential applications in chemistry, drug discovery, other areas of science and technology as well as in daily life activities. In this regard the advancements on the development of more sustainable and expedient as well as novel organic synthetic methodologies is of paramount importance. By using suitable catalysts (mentioned above) we perform advanced organic synthesis in a more sustainable, efficient and selective manner. In particular we focus on the development of sustainable and cost-effective processes for the synthesis of industrial fine and bulk chemicals, functionalized and structurally diverse molecules, pharmaceuticals and agrochemicals as well as for the utilization of renewable feedstocks (CO2 and biomass). For these syntheses, we use catalytic C-N, C-C, C-O and C-X (X=F, Cl and Br) bond forming and cleaving reactions. More specifically, we apply industrially relevant processes such as catalytic hydrogenations, aminations, oxidations, ammoxidations, dehydrogenations and hydrodeoxygenations, coupling reactions as well as CH-activation and functionalization. To perform these processes, we use inexpensive and easily accessible starting materials and renewable feedstocks as well as more abundant and green reagents.
Our selected publications related to these works: Science, 2022; 376, 1433-1441; Science, 2013, 342, 1073-1076; Science, 2017, 358, 326-332; Nature Catal., 2022, 5, 20-29; Chem, 2022, 8, 508-531.
Representative publications
1. V. G. Chandrashekhar, W. Baumann, M. Matthias, R. V. Jagadeesh, Science, 2022 (Accepted). Nickel-catalyzed hydrogenative coupling of nitriles and amines for general amine synthesis.
2. R. V. Jagadeesh, K. Murugesan, A. S. Alshammari, H. Neumann, M.-M. Pohl, J. Radnik, M. Beller, Science, 2017, 358, 326-332. MOF-derived cobalt nanoparticles catalyze a general synthesis of amines.
3. R. V. Jagadeesh, A. E. Surkus, H. Junge, M. M. Pohl, J. Radnik, J. Rabeah, H. Huan, V. Schünemann, A. Brückner, M. Beller, Science, 2013, 342, 1073-1076. Nanoscale Fe2O3-based catalysts for selective hydrogenation of nitroarenes to anilines.
4. V. G. Chandrashekhar, T. Senthamarai, R. G. Kadam, O. Malina, J. Kašlík, R. Zbořil, M. B. Gawande, R. V. Jagadeesh, M. Beller, Nature Catalysis, 2022, 5, 20-29. Silica supported Fe/Fe-O nanoparticles for the catalytic hydrogenation of nitriles to amines in the presence of aluminium additives.
5. T. Senthamarai, V. G. Chandrashekhar, N. Rockstroh, J. Rabeah, S. Bartling, R. V. Jagadeesh, M. Beller, Chem, 2022, 8, 508-531. A “universal” catalyst for aerobic oxidations to synthesize (hetero)aromatic aldehydes, ketones, esters, acids, nitriles and amides.
6. R. V. Jagadeesh, H. Junge, M. M. Pohl, J. Radnik, A. Brückner, M. Beller, Journal of the American Chemical Society, 2013, 135, 10776-10782. Selective oxidation of alcohols to esters using heterogeneous Co3O4-N@C under mild conditions.
7. K. Murugesan, Z. Wei, V. G. Chandrashekhar, H. Neumann, A. Spannenberg, H. Jiao, M. Beller, R. V. Jagadeesh, Nature Communications, 2019, 10, 5443. Homogeneous cobalt-catalyzed reductive amination for synthesis of functionalized primary amines.
8. T. Senthamarai, K. Murugesan, J. Schneidewind, N. V. Kalevaru, W. Baumann, H. Neumann, P. C. J. Kamer, M. Beller, R. V. Jagadeesh, Nature Communications, 2018, 9, 4123. Simple ruthenium-catalyzed reductive amination enables the synthesis of a broad range of primary amines.
9. K. Murugesan, V. G. Chandrashekhar, C. Kreyenschulte, M. Beller, R. V. Jagadeesh, Angewandte Chemie International Edition, 2020, 59, 17408-17412. A general catalyst based on cobalt-core-shell nanoparticles for hydrogenation of N-heteroarenes including pyridines.
10. J. Gao, L. Feng, R. Ma, B. J. Su, A. M. Alenad, Y. Liu, M. Beller, R. V. Jagadeesh, Chem Catalysis, 2022, 2, 178-194. Cobalt single-atom catalysts for domino reductive amination and amidation of levulinic acid and related molecules to N-heterocycles.


